新闻资讯
廖洪钢教授2023年论文汇总
2024-01-02
作者:
浏览量:
人物介绍

廖洪钢教授
廖洪钢,教授、博士生导师,国家高层次引进人才、闽江学者特聘教授。主要研究领域为原位电镜技术开发及其在材料合成、电催化、能源存储与转换过程的应用。完成了原位液体透射电镜的早期示范性研究,在Science、Nature及其子刊、JACS、Angew、EES等期刊发表论文100余篇,申请及授权专利、软著60余项,自主开发多种原位电镜芯片及分析系统,可在基础研究及产业升级等方面广泛应用。目前担任中国能源学会专家委员会委员,中国化学会高分子材料分析技术与表征方法专业委员会委员,厦门大学校友会创业分会监事长。
今天我们汇总盘点一下廖洪钢教授在2023年取得的重要研究成果。
2023
Sep
Nature
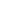
Visualizing interfacial collective reaction behaviour of Li–S batteries
Abstract
Benefiting from high energy density (2,600 Wh kg−1) and low cost, lithium–sulfur (Li–S) batteries are considered promising candidates for advanced energy-storage systems1,2,3,4. Despite tremendous efforts in suppressing the long-standing shuttle effect of lithium polysulfides5,6,7, understanding of the interfacial reactions of lithium polysulfides at the nanoscale remains elusive. This is mainly because of the limitations of in situ characterization tools in tracing the liquid–solid conversion of unstable lithium polysulfides at high temporal–spatial resolution8,9,10. There is an urgent need to understand the coupled phenomena inside Li–S batteries, specifically, the dynamic distribution, aggregation, deposition and dissolution of lithium polysulfides. Here, by using in situ liquid-cell electrochemical transmission electron microscopy, we directly visualized the transformation of lithium polysulfides over electrode surfaces at the atomic scale. Notably, an unexpected gathering-induced collective charge transfer of lithium polysulfides was captured on the nanocluster active-centre-immobilized surface. It further induced an instantaneous deposition of nonequilibrium Li2S nanocrystals from the dense liquid phase of lithium polysulfides. Without mediation of active centres, the reactions followed a classical single-molecule pathway, lithium polysulfides transforming into Li2S2 and Li2S step by step. Molecular dynamics simulations indicated that the long-range electrostatic interaction between active centres and lithium polysulfides promoted the formation of a dense phase consisting of Li+ and Sn2− (2 < n ≤ 6), and the collective charge transfer in the dense phase was further verified by ab initio molecular dynamics simulations. The collective interfacial reaction pathway unveils a new transformation mechanism and deepens the fundamental understanding of Li–S batteries.
Link:https://doi.org/10.1038/s41586-023-06326-8
Jan
Chemical Engineering Journal
In situ TEM observation of liquid flow induced dense phase formation and anisotropic nanocrystal growth
Abstract
The nucleation and growth of crystals from solutions are fundamental to materials synthesis and its applications. The mass transfer at micro/nano region in solution will greatly influence the crystal growth process, however, to our knowledge, there is rarely research at this scale due to the technique limit. By using in situ liquid cell transmission electron microscopy (TEM), we observed the crystal growth at flow condition with high resolution in real time. Interestingly, we found a flow induced formation of the solute rich dense liquid phase near surface at ultra-low precursor concentration, which then transformed to nanocrystals. The dendritic crystal growth was accelerated by the dense phase droplet merging and the anisotropic crystal growth was controlled by dense phase shape evolution. Our study deepens the understanding of crystal growth mechanisms, which is crucial to determine reaction dynamics and design hierarchical nanomaterials.
Link:https://doi.org/10.1016/j.cej.2022.140748
Jan
Chemical Science
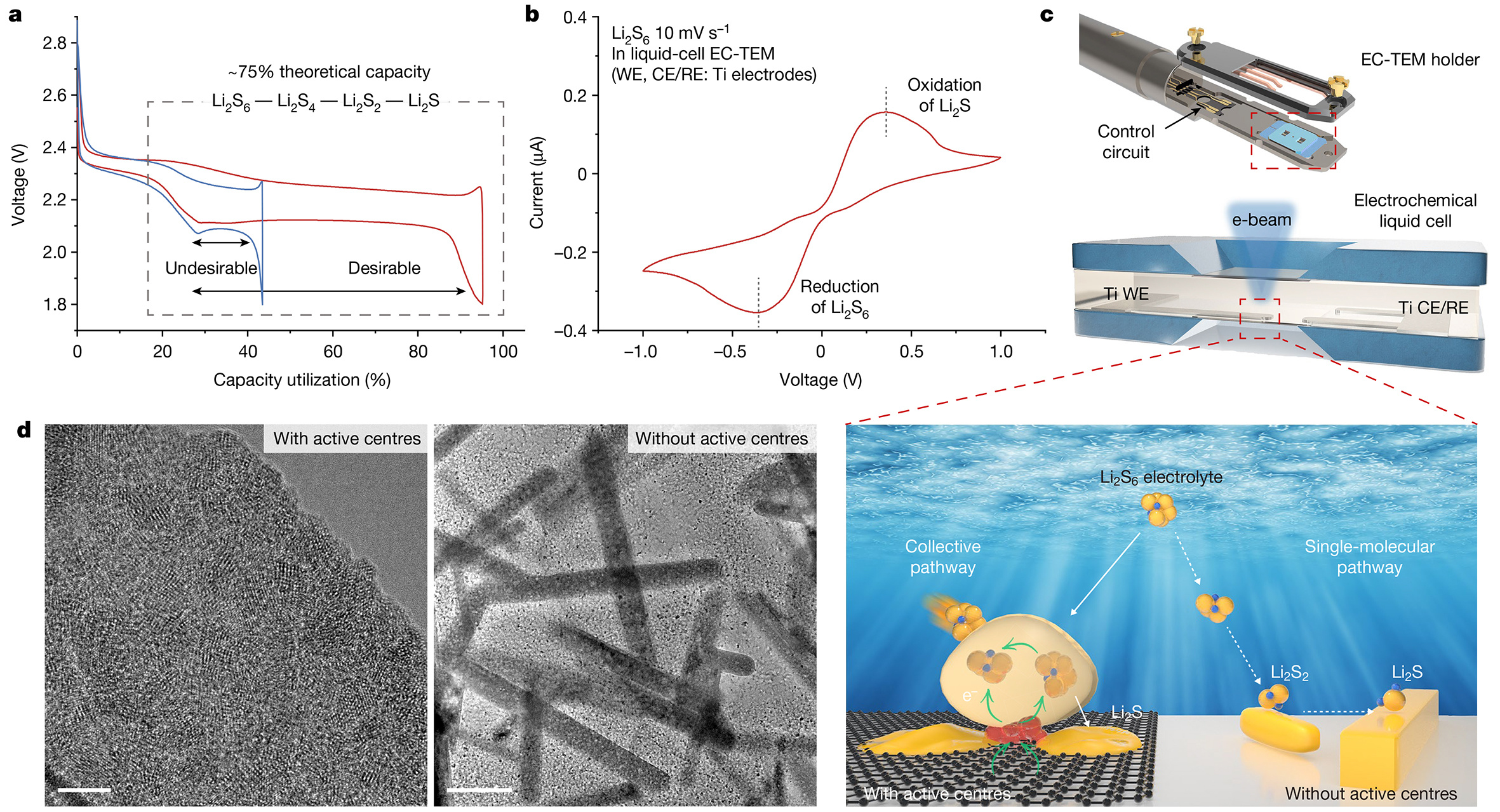
Prolonged lifespan of initial-anode-free lithium-metal battery by pre-lithiation in Li-rich Li2Ni0.5Mn1.5O4 spinel cathode
Abstract
Anode-free lithium metal batteries (AF-LMBs) can deliver the maximum energy density. However, achieving AF-LMBs with a long lifespan remains challenging because of the poor reversibility of Li+ plating/stripping on the anode. Here, coupled with a fluorine-containing electrolyte, we introduce a cathode pre-lithiation strategy to extend the lifespan of AF-LMBs. The AF-LMB is constructed with Li-rich Li2Ni0.5Mn1.5O4 cathodes as a Li-ion extender; the Li2Ni0.5Mn1.5O4 can deliver a large amount of Li+ in the initial charging process to offset the continuous Li+ consumption, which benefits the cycling performance without sacrificing energy density. Moreover, the cathode pre-lithiation design has been practically and precisely regulated using engineering methods (Li-metal contact and pre-lithiation Li-biphenyl immersion). Benefiting from the highly reversible Li metal on the Cu anode and Li2Ni0.5Mn1.5O4 cathode, the further fabricated anode-free pouch cells achieve 350 W h kg−1 energy density and 97% capacity retention after 50 cycles.
Link: https://doi.org/10.1039/d2sc06772b
Feb
Advanced Functional Materials
Restraining Shuttle Effect in Rechargeable Batteries by Multifunctional Zeolite Coated Separator

Abstract
The detrimental shuttle of soluble species from cathode to anode inside battery, is a critical thorn limiting stability and reversibility of rechargeable battery. Herein, an ordered pore-window of zeolite molecular sieve is employed to effectively block shuttle of soluble matters, and prepared zeolite powder into thin zeolite layer (5 µm thick) coated on celgard separator (zeolite@celgard) with flexible and grid-scale fabrication features. External pressure is applied to press zeolite@celgard to reduce existed interparticle gaps among zeolite particles. The separation function toward soluble species and attached H2O scavenger role of zeolite@celgard are demonstrated via 1H/19F Nuclear Magnetic Resonance spectra, Inductive Coupled Plasma Emission Spectrometer and X-Ray Photoelectron Spectroscopy results collected from Li/LiMn2O4 battery, time-dependent in situ Raman tests in Li/S battery, and penetration experiments of redox mediator shuttle in Li/O2 battery. Replacing typically-used celgard/glassfiber separators, a series of side reactions (active material outflowing, low coulombic efficiency, and anode corrosion) induced via shuttle of soluble species are addressed, resulting in battery performance improvement of Li/LiMn2O4, Li/S, and Li/O2 batteries. Both scientific hypothesis of utilizing pore-size effect of zeolite for physically block soluble species, and cost-effective, grid-scale, and flexible zeolite-based separators can be extended to other rechargeable battery systems based on flowing/soluble species.
Link: https://doi.org/10.1002/adfm.202211774
Mar
Communications Chemistry
Real-time imaging of sulfhydryl single-stranded DNA aggregation

Abstract
The structure and functionality of biomacromolecules are often regulated by chemical bonds, however, the regulation process and underlying mechanisms have not been well understood. Here, by using in situ liquid-phase transmission electron microscopy (LP-TEM), we explored the function of disulfide bonds during the self-assembly and structural evolution of sulfhydryl single-stranded DNA (SH-ssDNA). Sulfhydryl groups could induce self-assembly of SH-ssDNA into circular DNA containing disulfide bonds (SS-cirDNA). In addition, the disulfide bond interaction triggered the aggregation of two SS-cirDNA macromolecules along with significant structural changes. This visualization strategy provided structure information at nanometer resolution in real time and space, which could benefit future biomacromolecules research.
Link:https://doi.org/10.1038/s42004-023-00886-6
Mar
Molecules
Cerium Synchronous Doping in Anatase for Enhanced Photocatalytic Hydrogen Production from Ethanol-Water Mixtures

Abstract
Cerium element with a unique electric structure can be used to modify semiconductor photocatalysts to enhance their photocatalytic performances. In this work, Ce-doped TiO2 (Ce/TiO2) was successfully achieved using the sol-gel method. The structural characterization methods confirm that Ce was doped in the lattice of anatase TiO2, which led to a smaller grain size. The performance test results show that the Ce doped in anatase TiO2 significantly enhances the charge transport efficiency and broadens the light absorption range, resulting in higher photocatalytic performance. The Ce/TiO2 exhibited a photocurrent density of 10.9 μA/cm2 at 1.0 V vs. Ag/AgCl, 2.5 times higher than that of pure TiO2 (4.3 μA/cm2) under AM 1.5 G light. The hydrogen (H2) production rate of the Ce/TiO2 was approximately 0.33 μmol/h/g, which is more than twice as much as that of the pure anatase TiO2 (0.12 μmol/h/g). This work demonstrates the effect of Ce doping in the lattice of TiO2 for enhanced photocatalytic hydrogen production.
Link:https://doi.org/10.3390/molecules28062433
Mar
Advanced Materials
Construction of Metal–Organic Framework Films via Crosslinking-Induced Assembly

Abstract
The construction of metal–organic framework (MOF) films is a crucial step for integrating them into technical applications. However, due to the crystallization nature, it is difficult to grow most MOFs spontaneously or process them into films. Here, a convenient strategy is demonstrated for constructing MOF films by using modulators to achieve homogeneous assembly of MOF clusters. Small clusters in the early growth steps of MOFs can be stabilized by modulators to form fluidic precursors with good processibility. Then, simple removal of modulators will trigger the crosslinking of MOF clusters and lead to the formation of continuous films. This strategy is universal for the fabrication of several types of MOF films with large scale and controllable thickness, which can be deposited on a variety of substrates as well as can be patterned in micro/nano resolution. Additionally, versatile composite MOF films can be easily synthesized by introducing functional materials during the crosslinking process, which brings them broader application prospects.
Link:https://doi.org/10.1002/adma.202209777
Apr
Journal of the American Chemical Society
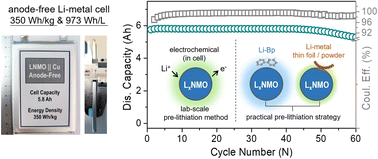
Role of Substitution Elements in Enhancing the Structural Stability of Li-Rich Layered Cathodes
Abstract
Element doping/substitution has been recognized as an effective strategy to enhance the structural stability of layered cathodes. However, abundant substitution studies not only lack a clear identification of the substitution sites in the material lattice, but the rigid interpretation of the transition metal (TM)–O covalent theory is also not sufficiently convincing, resulting in the doping/substitution proposals being dragged into design blindness. In this work, taking Li1.2Ni0.2Mn0.6O2 as a prototype, the intense correlation between the “disordered degree” (Li/Ni mixing) and interface-structure stability (e.g., TM–O environment, slab/lattice, and Li+ reversibility) is revealed. Specifically, the degree of disorder induced by the Mg/Ti substitution extends in the opposite direction, conducive to sharp differences in the stability of TM–O, Li+ diffusion, and anion redox reversibility, delivering fairly distinct electrochemical performance. Based on the established paradigm of systematic characterization/analysis, the “degree of disorder” has been shown to be a powerful indicator of material modification by element substitution/doping.
Link:https://doi.org/10.1021/jacs.3c01999
May
Angewandte Chemie International Edition
Unveiling Atom Migration Abilities Affected Anode Performance of Sodium-Ion Batteries
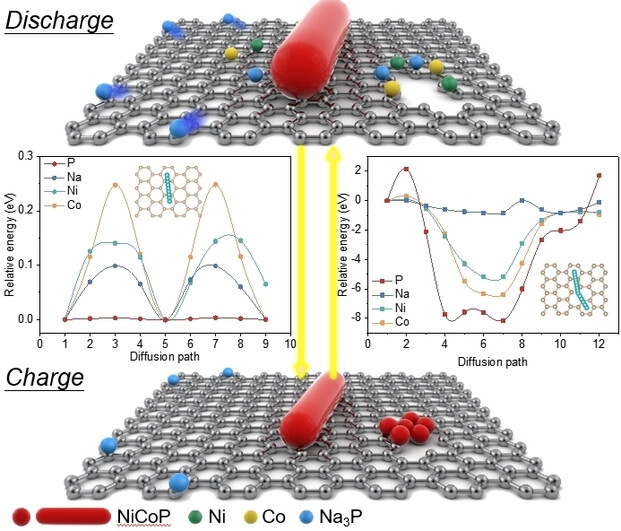
Abstract
In sodium-ion batteries (SIBs), the low initial coulombic efficiency (ICE) is commonly induced by irreversible phase conversion and difficult desodiation, especially on transition metal compounds (TMCs). Yet the underlying physicochemical mechanism of poor reaction reversibility is still a controversial issue. Herein, by using in situ transmission electron microscopy and in situ X-ray diffraction, we demonstrate the irreversible conversion of NiCoP@C is caused by the rapid migration of P in carbon layer and preferential formation of isolated Na3P during discharge. By modifying the carbon coating layer, the migration of Ni/Co/P atoms is inhibited, thus the improvement of ICE and cycle stability is realized. The inhibiting of fast atom migration which induces component separation and rapid performance degradation might be applied to a wide range of electrode materials, and guides the development of advanced SIBs.
Link: https://doi.org/10.1002/anie.202303343
May
Journal of Energy Chemistry
Spontaneous local redox reaction to passivate CNTs as lightweight current collector for high energy density lithium ion batteries
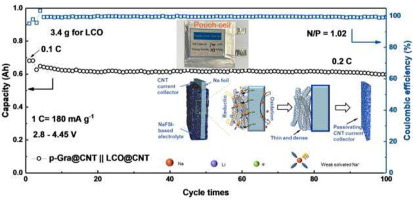
Abstract
Extensive usage of highly conductive carbon materials with large specific surface area (e.g., carbon nanotubes, CNTs) in lithium ion batteries (LIBs), especially as current collector of anodes, suffers from low initial coulombic efficiency (ICE), large interfacial resistance, and severe embrittlement, as the large specific surface area often results in severe interfacial decomposition of the electrolyte and the formation of thick and fluffy solid electrolyte interphase (SEI) during cycling of LIBs. Herein, we demonstrate that when the CNT-based current collector and Na foil (which are being stacked intimately upon each other) are being placed in Na+-based organic electrolyte, local redox reaction between the Na foil and the electrolyte would occur spontaneously, generating a thin and homogeneous NaF-based passivating layer on the CNTs. More importantly, we found that owing to the weak solvation behaviors of Na+ in the organic electrolyte, the resulting passivation layer, which is rich in NaF, is thin and dense; when used as the anode current collector in LIBs, the pre-existing passivating layer can function effectively in isolating the anode from the solvated Li+, thus suppressing the formation of bulky SEI and the destructive intercalation of solvated Li+. The relevant half-cell (graphite as anode) exhibits a high ICE of 92.1%; the relevant pouch cell with thus passivated CNT film as current collectors for both electrodes (LiCoO2 as cathode, graphite as anode) displays a high energy density of 255 Wh kg−1, spelling an increase of 50% compared with that using the conventional metal current collectors.
Link:https://doi.org/10.1016/j.jechem.2023.01.056
May
Journal of Materials Chemistry A
Enhancing the cycle-life of initial-anode-free lithium-metal batteries by pre-lithiation in Mn-based Li-rich spinel cathodes

Abstract
Anode-free lithium metal batteries (AF-LMBs) are attracting growing attention due to the burgeoning pursuit of high energy density. However, the poor reversibility of lithium-ion plating/stripping on the Cu anode triggers multiple issues, including rapid capacity loss, active lithium loss, and short lifetime. Herein, a cathode pre-lithiation strategy combined with a fluorine-containing electrolyte is introduced to achieve long-lifetime AF-LMBs. Furthermore, contact pre-lithiation, a more advanced and convenient strategy, has been introduced to precisely control the lithiation degree by adjusting the contact time. It has been demonstrated that over-lithiated Li1.3Mn2O4 can deliver additional lithium ions during the charging process, thus exhibiting 94.4% capacity retention after 40 cycles.
Link:https://doi.org/10.1039/d3ta01823g
Jun
Angewandte Chemie International Edition
Instantaneous Free Radical Scavenging by CeO2 Nanoparticles Adjacent to the Fe−N4 Active Sites for Durable Fuel Cells

Abstract
To achieve the Fe−N−C materials with both high activity and durability in proton exchange membrane fuel cells, the attack of free radicals on Fe−N4 sites must be overcome. Herein, we report a strategy to effectively eliminate radicals at the source to mitigate the degradation by anchoring CeO2 nanoparticles as radicals scavengers adjacent (Scaad-CeO2) to the Fe−N4 sites. Radicals such as ⋅OH and HO2⋅ that form at Fe−N4 sites can be instantaneously eliminated by adjacent CeO2, which shortens the survival time of radicals and the regional space of their damage. As a result, the CeO2 scavengers in Fe−NC/Scaad-CeO2 achieved ~80 % elimination of the radicals generated at the Fe−N4 sites. A fuel cell prepared with the Fe−NC/Scaad-CeO2 showed a smaller peak power density decay after 30,000 cycles determined with US DOE PGM-relevant AST, increasing the decay of Fe−NCPhen from 69 % to 28 % decay.
Link: https://doi.org/10.1002/ange.202306166
Jun
Materials Today Nano
Cycle-stable Si-based composite anode for lithium-ion batteries enabled by the synergetic combination of mixed lithium phosphates and void-preserving F-doped carbon

Abstract
Silicon-based materials have been considered as the promising anode candidates for next-generation high-energy-density lithium-ion batteries (LIBs). However, their widespread application is unfortunately restricted by severe volume variations during cycling and poor electronic conductivity. To overcome these challenges, we showcase an innovative design of dual core−shell structured Si-based nanocomposites working as anode for LIBs with boosted performance, where the Si nanoparticle core is tightly wrapped by a mixed lithium phosphate (Li3PO4/Li4P2O7) shell and void-preserving F-doped carbon shell (denoted as Si@LPO@void@FC). For such novel structured composite, the inner L3PO4/Li4P2O7 layer acting as artificial solid-electrolyte interphase (SEI) and the outer void-preserving F-doped C can effectively tolerate the volume changes while ensuring the stability of SEI layer, facilitate the Li+ migration and electron transfer, and reinforce the structural stability during cycling. Consequently, the as-fabricated Si@LPO@void@FC anode exhibits a reversible capacity of 569 mAh/g after 500 cycles at 1 A/g, and an exceptional long-term cycling stability with 76% capacity retention over 1000 cycles at 4.0 A/g can be achieved. Additionally, the full cell assembled with Si@LPO@void@FC anode and LiFePO4 cathode also demonstrates a good cycling performance with 117 mAh/g at 1 C for over 150 cycles with 92% capacity retention, suggesting the potentiality for practical application. Furthermore, the mechanisms of the enhanced structural stability of Si@LPO@void@FC anode are carefully elaborated by substantial in situ/ex situ microscopic techniques and electrochemical tests. It is expected that our findings in this work can provide guiding significance for improving the cycling performance of Si-based composite anodes toward high-performance LIBs.
Link:https://doi.org/10.1016/j.mtnano.2023.100322
Jul
Royal Society of Chemistry
On-chip gas reaction nanolab for in situ TEM observation

Abstract
The catalysis reaction mechanism at nano/atomic scale attracted intense attention in the past decades. However, most in situ characterization technologies can only reflect the average information of catalysts, which leads to the inability to characterize the dynamic changes of single nanostructures or active sites under operando conditions, and many micro–nanoscale reaction mechanisms are still unknown. The combination of in situ transmission electron microscopy (TEM) holder system with MEMS chips provides a solution for it, where the design and fabrication of MEMS chips are the key factors. Here, with the aid of finite element simulation, an ultra-stable heating chip was developed, which has an ultra-low thermal drift during temperature heating. Under ambient conditions within TEM, atomic resolution imaging was achieved during the heating process or at high temperature up to 1300 °C. Combined with the developed polymer membrane seal technique and nanofluidic control system, it can realize an adjustable pressure from 0.1 bar to 4 bar gas environment around the sample. By using the developed ultra-low drift gas reaction cells, the nanoparticle's structure evolution at atomic scale was identified during reaction.
Link:https://doi.org/10.1039/D3LC00184A
Jul
Science Advances
Observing ion diffusion and reciprocating hopping motion in water

Abstract
When an ionic crystal dissolves in solvent, the positive and negative ions associated with solvent molecules release from the crystal. However, the existing form, interaction, and dynamics of ions in real solution are poorly understood because of the substantial experimental challenge. We observed the diffusion and aggregation of polyoxometalate (POM) ions in water by using liquid phase transmission electron microscopy. Real-time observation reveals an unexpected local reciprocating hopping motion of the ions in water, which may be caused by the short-range polymerized bridge of water molecules. We find that ion oligomers, existing as highly active clusters, undergo frequent splitting, aggregation, and rearrangement in dilute solution. The formation and dissociation of ion oligomers indicate a weak counterion-mediated interaction. Furthermore, POM ions with tetrahedral geometry show directional interaction compared with spherical ions, which presents structure-dependent dynamics.
Link:https://www.science.org/doi/10.1126/sciadv.adf8436
Sep
Nano Energy
In-situ constructing Bi@Bi2O2CO3 nanosheet catalyst for ampere-level CO2 electroreduction to formate
Abstract
Bi-based electrocatalysts are prominent candidates to achieve CO2 reduction to formate with high selectivity but suffer from unsatisfied activity, stability, and ambiguous nature of active sites. Herein, the Bi@Bi2O2CO3 nanosheet catalyst is designed via an electrochemical in-situ reconstruction approach from BiPO4. The conversion processes were revealed by electrochemical in-situ Raman and FTIR spectroscopy. The obtained Bi@Bi2O2CO3 catalyst demonstrates high CO2 reduction performance (FEHCOOH ≈ 100%, jHCOOH ≈ −60 mA cm−2) and excellent stability of 110 h in H-type cell. Moreover, the Bi@Bi2O2CO3 delivers a remarkable formate partial current density up to −1.2 A cm−2 (production rate as 22.4 mmol cm−2 h−1) in the flow cell. DFT theoretical studies reveal the synergistic effect of Bi and Bi2O2CO3 at the interface played an important role in changing the adsorption behavior of reaction intermediates and further greatly reducing the activation barrier of the conversion of *OCHO to *HCOOH during CO2 reduction.
Link:https://doi.org/10.1016/j.nanoen.2023.108638
Oct
Current Opinion in Electrochemistry
Perspective of operando/in situ scanning electron microscope in rechargeable batteries

Abstract
With an ever-increasing demand for clean energy, rechargeable batteries have undergone significant development, owing to their high energy density, power density, safety, and affordability. It is crucial to employ qualitative and quantitative techniques that can operate under real battery working conditions. These techniques enable the determination of the dynamic evolution of the microstructure, phase composition, chemical composition, and local chemical environment of the battery interior and interface. Operando/in situ scanning electron microscopy (SEM) has emerged as a valuable tool in rechargeable battery research, benefited from its high temporo-spatial resolution and comprehensive analysis capabilities facilitated by multiple detectors. It has found widespread application in understanding the fundamental mechanisms and real-time tracking of reaction dynamics in rechargeable batteries. This concise review focuses specifically on operando/in situ SEM with various configurations for investigating interfacial phenomena in rechargeable batteries. Furthermore, notable future opportunities and developments have been discussed.
Link:https://doi.org/10.1016/j.coelec.2023.101374
Nov
Nature Protocols
Fabrication of liquid cell for in situ transmission electron microscopy of electrochemical processes

Abstract
Fundamentally understanding the complex electrochemical reactions that are associated with energy devices (e.g., rechargeable batteries, fuel cells and electrolyzers) has attracted worldwide attention. In situ liquid cell transmission electron microscopy (TEM) offers opportunities to directly observe and analyze in-liquid specimens without the need for freezing or drying, which opens up a door for visualizing these complex electrochemical reactions at the nano scale in real time. The key to the success of this technique lies in the design and fabrication of electrochemical liquid cells with thin but strong imaging windows. This protocol describes the detailed procedures of our established technique for the fabrication of such electrochemical liquid cells (~110 h). In addition, the protocol for the in situ TEM observation of electrochemical reactions by using the nanofabricated electrochemical liquid cell is also presented (2 h). We also show and analyze experimental results relating to the electrochemical reactions captured. We believe that this protocol will shed light on strategies for fabricating high-quality TEM liquid cells for probing dynamic electrochemical reactions in high resolution, providing a powerful research tool. This protocol requires access to a clean room equipped with specialized nanofabrication setups as well as TEM characterization equipment.
Link:https://doi.org/10.1038/s41596-022-00762-y
暂无数据