新闻资讯
成果分享|廖洪钢教授2022年论文汇总
2023-02-01
作者:
浏览量:

廖洪钢,厦门大学化学系教授,国家高层次引进人才、闽江学者特聘教授,主要研究领域为原位电镜技术开发及其在材料合成、催化、能源存储与转换过程的应用。
完成了原位液体透射电镜的早期示范性研究,研究工作发表在2012及2014《Science》等刊物上,被报道和评论为“塑造纳米晶体的未来”,“颠覆了一百多年来对晶体生长规律的认知”。
2015年回国后,打破国外垄断,自主开发成功多种原位电镜分析芯片及系统,共申请授权专利、软著近百项,可在基础研究及产业升级等方面广泛应用。累计已在Science,Nature子刊等SCI期刊上发表论文80余篇。
今天我们汇总盘点一下廖洪钢教授在2022年取得的重要研究成果。并且,从本期开始,将陆续对这些研究成果做介绍分享。
2022
2022 Jan
Energy & Environmental Science:Efficient diffusion of superdense lithium via atomic channels for dendrite-free lithium–metal batteries
 The non-uniform aggregation of fast-diffused Li on an anode surface would aggravate its tip-effect-induced nucleation/growth, leading to the notorious dendrite growth in Li metal batteries (LMBs). Tuning the Li diffusion on the anode surface has been regarded previously as a mainstream method to induce its uniform deposition, while the diffusion of Li in the anode bulk is usually ignored. Here, conceptually different from the classic surface modification, we propose a molecular tunnelling strategy to construct atomic channels in graphite bulk, which enables the fast diffusion of superdense Li. Density functional theory calculations and ab initio molecular dynamics simulations prove that the bulk diffusion through atomic channels could become a new and dominating path. Its reversible and efficient diffusion has been further visualized by in situ transmission electron microscopy. As a result, when coupled with high-loading LiFePO4 cathodes (20 mg cm닋2), a high areal capacity and 100% capacity retention are achieved over 370 cycles. Through this work a new strategy is developed based on the bulk-diffusion of superdense Li for dendrite-free LMBs, which can be pervasive in other high-performance energy storage systems.
The non-uniform aggregation of fast-diffused Li on an anode surface would aggravate its tip-effect-induced nucleation/growth, leading to the notorious dendrite growth in Li metal batteries (LMBs). Tuning the Li diffusion on the anode surface has been regarded previously as a mainstream method to induce its uniform deposition, while the diffusion of Li in the anode bulk is usually ignored. Here, conceptually different from the classic surface modification, we propose a molecular tunnelling strategy to construct atomic channels in graphite bulk, which enables the fast diffusion of superdense Li. Density functional theory calculations and ab initio molecular dynamics simulations prove that the bulk diffusion through atomic channels could become a new and dominating path. Its reversible and efficient diffusion has been further visualized by in situ transmission electron microscopy. As a result, when coupled with high-loading LiFePO4 cathodes (20 mg cm닋2), a high areal capacity and 100% capacity retention are achieved over 370 cycles. Through this work a new strategy is developed based on the bulk-diffusion of superdense Li for dendrite-free LMBs, which can be pervasive in other high-performance energy storage systems.
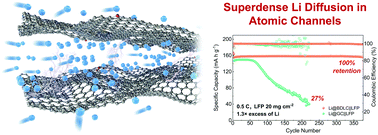
Link:https://doi.org/10.1039/D1EE02205A
2022 Mar
Journal of Materials Chemistry A:Co/Li-dual-site doping towards LiCoO2 as a high-voltage, fast-charging, and long-cycling cathode material

The design of fast-charging, long-cycling, and high-voltage cathode materials remains challenging. Herein, through different strategies, Al and Nb/W are doped into the Co- and Li-sites in LiCoO2 (LCO), respectively; according to density functional theory calculations, compared with the Co-site, doping at Li-site is thermodynamically unfavourable, which is primarily driven by the kinetic motif. We demonstrate that the Al-dopant at the Co-site inhibits the adverse phase transformation of LiCoO2 under high voltage, while the Nb/W dopants intercalated within the Li-slab can serve as pillars that not only increase the interlayer spacing but also decrease the electronic coupling around Li+, thus increasing the population of highly active Li+ and enabling fast Li+ diffusion kinetics. Owing to the synergy effect from dual-site doping at both Co- and Li-sites, together with a discrete coating layer of niobium tungsten oxide (NWO) nanoparticles, the thus modified LiCoO2 (denoted as ANW-LCO) cathode delivers highly stable and superior rate performance even under high voltage. Specifically, with a cut-off potential of 4.5 V, it displays a specific capacity of as high as 142.1 mA h g−1 at 15C and can maintain a reversible capacity of 85.3 mA h g−1 after 1000 cycles at 10C under 4.5 V, translating into a capacity retention of 60.4%. When evaluated at 4.6 V, it shows a capacity retention of as high as 77.5% after 100 cycles. When tested in all-solid-state lithium-ion batteries, it delivers a primal discharge specific capacity of 139 mA h g−1 and retains 71% of its capacity after 200 cycles. The full-cell also demonstrates outstanding cycling stability, with a capacity retention of 71% after 500 cycles at 2C.
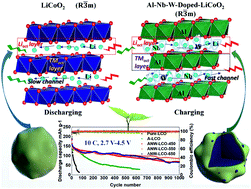
Link:https://doi.org/10.1039/D1TA10612K
2022 May
Science Bulletin:Boosting the potassium-ion storage performance enabled by engineering of hierarchical MoSSe nanosheets modified with carbon on porous carbon sphere

Developing suitable electrode materials capable of tolerating severe structural deformation and overcoming sluggish reaction kinetics resulting from the large radius of potassium ion (K+) insertion is critical for practical applications of potassium-ion batteries (PIBs). Herein, a superior anode material featuring an intriguing hierarchical structure where assembled MoSSe nanosheets are tightly anchored on a highly porous micron-sized carbon sphere and encapsulated within a thin carbon layer (denoted as Cs@MoSSe@C) is reported, which can significantly boost the performance of PIBs. The assembled MoSSe nanosheets with expanded interlayer spacing and rich anion vacancy can facilitate the intercalation/deintercalation of K+ and guarantee abundant active sites together with a low K+ diffusion barrier. Meanwhile, the thin carbon protective layer and the highly porous carbon sphere matrix can alleviate the volume expansion and enhance the charge transport within the composite. Under these merits, the as-prepared Cs@MoSSe@C anode exhibits a high reversible capacity (431.8 mAh g−1 at 0.05 A g−1), good rate capability (161 mAh g−1 at 5 A g−1), and superior cyclic performance (70.5% capacity retention after 600 cycles at 1 A g−1), outperforming most existing Mo-based S/Se anodes. The underlying mechanisms and origins of superior performance are elucidated by a set of correlated in-situ/ex-situ characterizations and theoretical calculations. Further, a PIB full cell based on Cs@MoSSe@C anode also exhibits an impressive electrochemical performance. This work provides some insights into developing high-performance PIBs anodes with transition-metal chalcogenides.
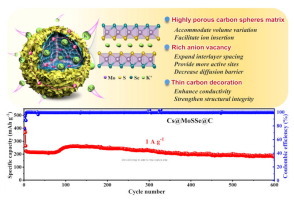
Link:https://doi.org/10.1016/j.scib.2022.02.007
2022 Jun
Applied Catalysis B: Environmental:Efficient CO2 reduction MOFs derivatives transformation mechanism revealed by in-situ liquid phase TEM

Materials derived from MOFs have great potentials in energy conversion. However, the nanoscale transformation processes of MOFs derivatives remain unknown. Herein, by using in-situ liquid phase TEM, we directly visualize the MOFs etching processes. For the first time, unexpected nanobubble stability controlled transformation mechanism of ZIF-67 to porous or layered cobalt transition metal hydroxide (Co-TMH) is identified. Voids in MOFs migrate and merge to form nanobubbles due to structural collapse. Under slow diffusion conditions, nanobubbles move slowly and Co-TMH clusters generate on the nanobubble interface, further favoring the formation of internal nanocages and porous structures. On the other hand, a fast diffusion leads to rapid nanobubbles generation, aggregation and reshaping, inducing layered structure formation. Inspired by in-situ observation, we further synthesize porous Co-TMH at -80 degrees C under inhibited diffusion conditions, which exhibits excellent catalytic performance on CO2 reduction reaction.
Link:https://doi.org/10.1016/j.apcatb.2022.121164
2022 Jun
NATURE COMMUNICATIONS:Identification of a quasi-liquid phase at solid–liquid interface

An understanding of solid–liquid interfaces is of great importance for fundamental research as well as industrial applications. However, it has been very challenging to directly image solid–liquid interfaces with high resolution, thus their structure and properties are often unknown. Here, we report a quasi-liquid phase between metal (In, Sn) nanoparticle surfaces and an aqueous solution observed using liquid cell transmission electron microscopy. Our real-time high-resolution imaging reveals a thin layer of liquid-like materials at the interfaces with the frequent appearance of small In nanoclusters. Such a quasi-liquid phase serves as an intermediate for the mass transport from the metal nanoparticle to the liquid. Density functional theory-molecular dynamics simulations demonstrate that the positive charges of In ions greatly contribute to the stabilization of the quasi-liquid phase on the metal surface.
Link:https://doi.org/10.1038/s41467-022-31075-z
2022 Jul
Energy & Environmental Scienc:Seizing gaseous Fe2+ to densify O2-accessible Fe–N4 sites for high-performance proton exchange membrane fuel cells

Increasing the density of Fe–N4 sites in Fe–N–C materials is pivotal for enhancing the kinetics of the oxygen reduction reaction (ORR) in proton exchange membrane fuel cells (PEMFCs). Fe utilization is a vital parameter for the Fe–N–C catalyst evaluation, but it shows a tendency to decrease with increasing density of the Fe–N4 sites. Herein, dense edge Fe–N2+2 sites are deposited in the outermost and subsurface layers of a surface-rich pyridinic-N carbon substrate (Feg–NC/Phen). We have demonstrated that the surface-rich pyridinic-N carbon substrate is more favorable to form surface Fe–N2+2 sites with superior intrinsic activity. The surface Fe–N4 sites can improve both the site density and Fe utilization, while shortening the transport pathways of protons and O2 effectively. By means of these structural advantages, Feg–NC/Phen can exhibit a high current density of 0.046 A cm−2@0.9 ViR-free and a high peak power density (Pmax) of 1.53 W cm−2 in 2 bar H2–O2 PEMFCs, and outperform almost all the reported M–N–C catalysts. This outstanding performance will inspire relevant research in the distribution of active sites. Moreover, it requires particular attention to obtain a viable solution to performance durability in fuel cells.
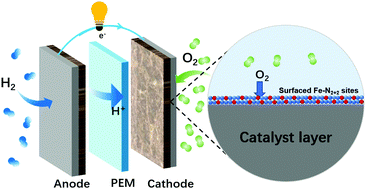
Link:https://doi.org/10.1039/D2EE00061J
2022 Jul
Journal of Materials Chemistry A:A superior electrocatalyst toward the oxygen reduction reaction obtained by atomically dispersing copper on N, F co-doped graphene through atomic interface engineering

Single-atom catalysts, featuring 100% metal atom utilization, strong metal–support interaction, low coordination environment, and high selectivity, have attracted great attention towards the enhancement of the oxygen reduction reaction (ORR). In the current work, we propose an atomic interface strategy for the construction of Cu single-atom catalysts dispersed on N, F co-doped graphene (Cu SA-NFG). The Cu SA-NFG catalyst exhibited excellent ORR activity in alkaline media. Both X-ray absorption fine structure and density functional theory results demonstrated that the Cu–N4 atomic interface is the active site for the ORR, and the electron-withdrawing F atoms can decrease the Gibbs free energy for the adsorption of intermediates. In addition, the Pt nanoparticle-decorated Cu SA-NFG catalyst displayed superior catalytic performance toward methanol oxidation. This work provides a new method for the rational design of advanced electrode materials and a new guide for the improvement of catalytic performance toward direct methanol fuel cells.
Link:https://doi.org/10.1039/D2TA01990F
2022 Sep
NATURE COMMUNICATIONS:Observation of formation and local structures of metal-organic layers via complementary electron microscopy techniques

Metal-organic layers (MOLs) are highly attractive for application in catalysis, separation, sensing and biomedicine, owing to their tunable framework structure. However, it is challenging to obtain comprehensive information about the formation and local structures of MOLs using standard electron microscopy methods due to serious damage under electron beam irradiation. Here, we investigate the growth processes and local structures of MOLs utilizing a combination of liquid-phase transmission electron microscopy, cryogenic electron microscopy and electron ptychography. Our results show a multistep formation process, where precursor clusters first form in solution, then they are complexed with ligands to form non-crystalline solids, followed by the arrangement of the cluster-ligand complex into crystalline sheets, with additional possible growth by the addition of clusters to surface edges. Moreover, high-resolution imaging allows us to identify missing clusters, dislocations, loop and flat surface terminations and ligand connectors in the MOLs. Our observations provide insights into controllable MOL crystal morphology, defect engineering, and surface modification, thus assisting novel MOL design and synthesis.
Link:https://doi.org/10.1038/s41467-022-32330-z
2022 Sep
Nano Letters:In Situ TEM Observation of Stagnant Liquid Layer Activation in Nanochannel

The kinetics of mass transfer in a stagnant fluid layer next to an interface govern numerous dynamic reactions in diffusional micro/nanopores, such as catalysis, fuel cells, and chemical separation. However, the effect of the interplay between stagnant liquid and flowing fluid on the micro/nanoscopic mass transfer dynamics remains poorly understood. Here, by using liquid cell transmission electron microscopy (TEM), we directly tracked microfluid unit migration at the nanoscale. By tracking the trajectories, an unexpected mass transfer phenomenon in which fluid units in the stagnant liquid layer migrated two orders faster during gas–liquid interface updating was identified. Molecular dynamics (MD) simulations indicated that the chemical potential difference between nanoscale liquid layers led to convective flow, which greatly enhanced mass transfer on the surface. Our study opens up a pathway toward research on mass transfer in the surface liquid layers at high spatial and temporal resolutions.
Link:https://doi.org/10.1021/acs.nanolett.2c01762
2022 Nov
ADVANCED FUNCTIONAL MATERIALS:Controlling Interfacial Structural Evolution in Aqueous Electrolyte via Anti-Electrolytic Zwitterionic Waterproofing

Aqueous electrolytes are considered as an alternative to flammable and toxic organic electrolytes, whose broad applications in electrochemical energy storage (EES) devices unfortunately suffer from low electrochemical stability due to the easy electrolysis of water. Here, by performing in situ transmission electron microscope electrochemical characterizations at atomic resolution during charging/discharging, an anti-electrolytic strategy is revealed in aqueous electrolytes via physical zwitterionic waterproofing. It is found that the zwitterionic molecules can be directionally adsorbed to the negative electrode's surface under the applied electric field, forming strings of zwitterionic molecules that extract water out from the electrode. More zwitterionic molecules further aggregate at the outer end of the strings through intermolecular electrostatic interactions, forming a waterproof layer that successfully expels water from the electrode's surface. Meanwhile, the self-aggregation of zwitterionic additives in the bulk liquid successfully minimizes the influence on ion transport. Being intrinsically distinct from the solid electrolyte interphase concept associated with certain electrochemical reactions in organic or super-concentrated electrolytes, the strategy is effective in improving the electrochemical stability while maintaining high ionic conductivity in various aqueous electrolytes even with a dilute concentration, shedding light on developing sustainable EES devices with high performance.
Link:https://doi.org/10.1002/adfm.202207140
2022 Nov
Nature Protocols:Fabrication of liquid cell for in situ transmission electron microscopy of electrochemical processes
Fundamentally understanding the complex electrochemical reactions that are associated with energy devices (e.g., rechargeable batteries, fuel cells and electrolyzers) has attracted worldwide attention. In situ liquid cell transmission electron microscopy (TEM) offers opportunities to directly observe and analyze in-liquid specimens without the need for freezing or drying, which opens up a door for visualizing these complex electrochemical reactions at the nano scale in real time. The key to the success of this technique lies in the design and fabrication of electrochemical liquid cells with thin but strong imaging windows. This protocol describes the detailed procedures of our established technique for the fabrication of such electrochemical liquid cells (~110 h). In addition, the protocol for the in situ TEM observation of electrochemical reactions by using the nanofabricated electrochemical liquid cell is also presented (2 h). We also show and analyze experimental results relating to the electrochemical reactions captured. We believe that this protocol will shed light on strategies for fabricating high-quality TEM liquid cells for probing dynamic electrochemical reactions in high resolution, providing a powerful research tool. This protocol requires access to a clean room equipped with specialized nanofabrication setups as well as TEM characterization equipment.
Link:https://doi.org/10.1038/s41596-022-00762-y
2022 Dec
Journal of Chemical Physics:Liquid cell electrochemical TEM: Unveiling the real-time interfacial reactions of advanced Li-metal batteries

Li metal batteries (LMBs) reveal great application prospect in next-generation energy storage, because of their high energy density and low electrochemical potential, especially when paired with elemental sulfur and oxygen cathodes. Complex interfacial reactions have long been a big concern because of the elusive formation/dissolution of Li metal at the solid–electrolyte interface (SEI) layer, which leads to battery degradation under practical operating conditions. To precisely track the reactions at the electrode/electrolyte interfaces, in the past ten years, high spatio–temporal resolution, in situ electrochemical transmission electron microscopy (EC-TEM) has been developed. A preliminary understanding of the structural and chemical variation of Li metal during nucleation/growth and SEI layer formation has been obtained. In this perspective, we give a brief introduction of liquid cell development. Then, we comparably discuss the different configurations of EC-TEM based on open-cell and liquid-cell, and focus on the recent advances of liquid-cell EC-TEM and its investigation in the electrodes, electrolytes, and SEI. Finally, we present a perspective of liquid-cell EC-TEM for future LMB research.
Link:https://doi.org/10.1063/5.0129238
2022 Dec
Chemical Engineering Journal:In situ TEM observation of liquid flow induced dense phase formation and anisotropic nanocrystal growth

The nucleation and growth of crystals from solutions are fundamental to materials synthesis and its applications. The mass transfer at micro/nano region in solution will greatly influence the crystal growth process, however, to our knowledge, there is rarely research at this scale due to the technique limit. By using in situ liquid cell transmission electron microscopy (TEM), we observed the crystal growth at flow condition with high resolution in real time. Interestingly, we found a flow induced formation of the solute rich dense liquid phase near surface at ultra-low precursor concentration, which then transformed to nanocrystals. The dendritic crystal growth was accelerated by the dense phase droplet merging and the anisotropic crystal growth was controlled by dense phase shape evolution. Our study deepens the understanding of crystal growth mechanisms, which is crucial to determine reaction dynamics and design hierarchical nanomaterials.
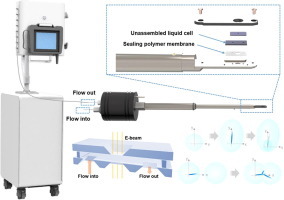
Link:https://doi.org/10.1016/j.cej.2022.140748
暂无数据

